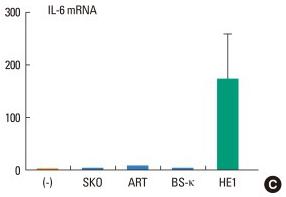
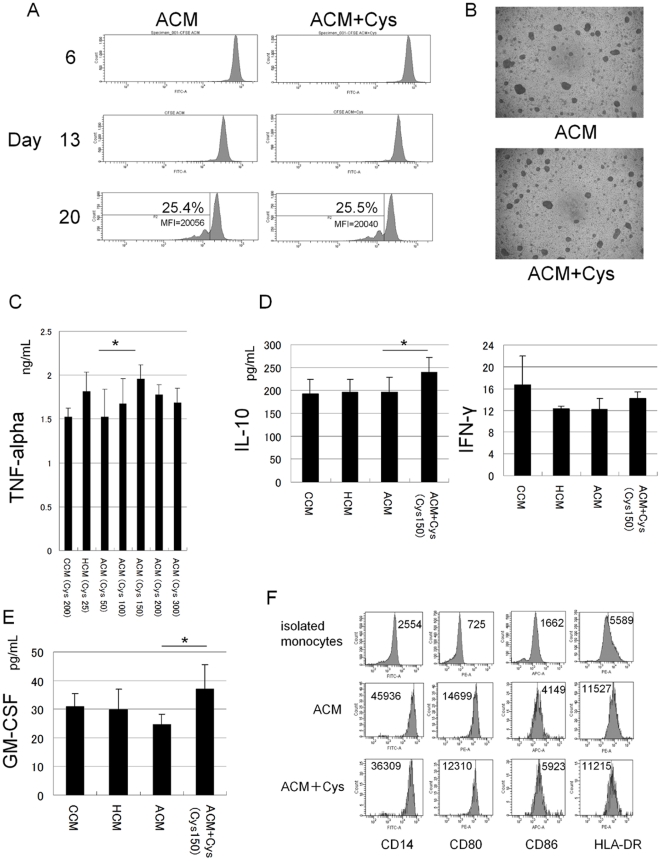
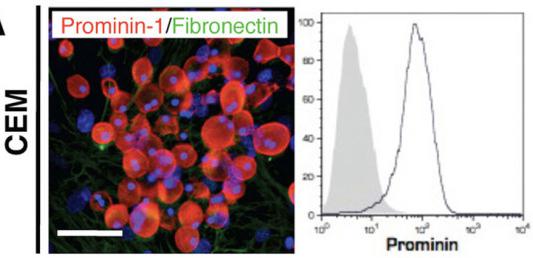
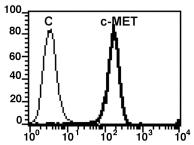
Human Stem Cell Factor Recombinant
Categories: PDGF familyPDGF familyRecombinant Human Cytokines$70.00 – $2,700.00
Description
Accession
P21583
Source
Optimized DNA sequence encoding Human SCF mature chain was expressed in Escherichia Coli.
Molecular weight
Native human Stem Cell Factor is generated by the proteolytic removal of the signal peptide and propeptide, the molecule has a calculated molecular mass of approximately 18 kDa. Recombinant SCF is a monomer protein consisting of 165 amino acid residue subunits, and migrates as an approximately 18 kDa protein under non-reducing and reducing conditions in SDS-PAGE.
Purity
>97%, as determined by SDS-PAGE and HPLC
Biological Activity
The ED(50) was determined by the dose-dependent stimulation of the proliferation of human TF-1 cells is ≤.0 ng/ml, corresponding to a specific activity of ≥ x units/mg.
Protein Sequence
MKKTQTWILT CIYLQLLLFN PLVKTEGICR NRVTNNVKDV TKLVANLPKD YMITLKYVPG MDVLPSHCWI SEMVVQLSDS LTDLLDKFSN ISEGLSNYSI IDKLVNIVDD LVECVKENSS KDLKKSFKSP EPRLFTPEEF FRIFNRSIDA FKDFVVASET SDCVVSSTLS PEKDSRVSVT KPFMLPPVA A SSLRNDSSSS NRKAKNPPGD SSLHWAAMAL PALFSLIIGF AFGALYWKKR QPSLTRAVEN IQINEEDNEI SMLQEKEREF QEV
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant SCF was lyophilized from a 0.2 μm filtered solution in PBS pH7.0.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at2° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Biological Process
Molecular function
Methods
Cell transfection and plasmids
- Human immature thymocytes were obtained as previously described + cells followed by negative selection using the human CD4+ T-cell enrichment cocktail according to the manufacturer's instructions.
- The Tax expression plasmid, pCMV-TaxGFP (generous gift from R. Mahieux), contains the
tax coding sequence in frame with the N-terminal extremity from thegfp sequence under the control of the cytomegalovirus promoter. - pMax-GFP was used as a GFP-control expressing vector.
- Immature celltype'>thymocytes (2.106 celltype'>cells), cultured overnight in cellcellmedium'>medium'>α-MEM cellcellmedium'>medium'>complete cellmedium'>medium containing 20 ng/ml IL-7 and 10 ng/ml SCF (ocky , ), were transfected either with 4 µg of pCMV-TaxGFP or pCMV-GFP by nucleofection (Human celltype'>C34 cell nucleofector kit, , ) according to the manufacturer's instructions.
- Twenty-four hours later, GFP+ cells were sorted from both cultures using an ARIA sorter (BD-Beckinson).
Processing whole blood samples
- Peripheral (PB.1 and PB.2) and cord (CB.1 and CB.2) blood-derived CD34+ cells were obtained from AllCells .
- Blood collections were performed at AllCells and using standard, 8 ml Vacutainer Cell Processing Tubes (both sodium citrate and sodium heparin-based tubes are acceptable ; , ).
- Appropriate documentation for informed consent was completed prior to blood collection .
- Vacutainers were processed within 24 hours of collection.
- Briefly, the PBMC-containing upper phase was collected and washed with ice-cold PBS .
- Cells were either frozen down or used directly for purification with the CD34 MicroBead Kit and used according to the manufacturer's protocol.
- Some samples were treated with Histopaque ( ; St. Louis, ) to minimize the number of red blood cells and centrifuged at 2000 rpm for 20 minutes without braking.
- The interface containing the PBMCs was removed if samples were treated with histopaque, cells washed again with chilled…
Erythroid precursor cultures
- After informed consent had been obtained, peripheral blood from CDA II patients and from 5 healthy control relatives was collected into sterile heparinised tubes.
- Light-density mononuclear cells obtained by centrifugation on density gradient were enriched for CD34+ cells by positive selection using CD34 microbeads according to the manufacturers' instructions.
- CD34+ cells were cultured at a density of 105 cells/mL in medium'>alpha-minimal medium'>essential medium (α-MEM; GIBCO) supplemented with 30% fetal bovine serum (FBS; GIBCO), as previously described [2 for 14 days; after 7 days of culture the medium was changed to ensure good cells feeding.
- Cell samples were collected on days 14 of culture (mature erythroblast stage) for further analysis.
2.3. Differentiation of hESC
-
H1 hESCs were plated at 3 × 106 per well of 6-well ultralow attachment (ULA) plates in a total volume of 4 mL of medium'>X-VIVO-15 medium , supplemented with 1 mM sodium pyruvate, nonessential amino acids, 2 mM L-glutamine (all PAA Laboratories GmbH) and 5
μ M 2-mercaptoethanol . - The following growth factors were added: 50 ng/mL recombinant human bone morphogenetic protein-4 (BMP-4, & ), 50 ng/mL recombinant human vascular endothelial growth factor , 20 ng/mL recombinant human stem cell factor , and 50 ng/mL recombinant human granulocyte macrophage-colony stimulating factor (GM-CSF, & ).
- After 2-3 days, the medium was topped up with 2 mL of fresh supplemented X-VIVO-15 medium to produce a total volume of 6 mL.
- Subsequent feeding was performed every 2-3 days by replacing 2-3 mL of old medium with new supplemented X-VIVO-15 medium from which every 5 days a growth factor was removed starting with BMP-4 at day 5, followed by VEGF at…
Cell culture
-
Primary human cord blood-derived mast cells (CBMC) were generated from umbilical cord blood mononuclear cells using a modification of the method of Saito
et al. and characterized as previously described 6 cells/mL and passaged weekly in RPMI-1640 medium supplemented with 20% FCS, 100 U/mL of penicillin, 100 µg/mL of streptomycin, 20% CCL-204 cell supernatant as a source of interleukin-6, 10−7 M prostaglandin E2 , and 50 to 100 ng/ml of stem cell factor . - For qPCR studies, CBMC cultures were sorted on a after at least 6 weeks of culture to enrich for high side scatter cells characteristic of mature mast cells.
- The sorted cells were returned to culture and the purity was assessed as described below.
- The purity of all cultures used was ≥95% as assessed by toluidine blue (pH 1.0) staining of cytocentrifuge preparations and examination of cells for the presence of multiple metachromatic granules and appropriate…
Cell Culture
- Frozen female CD34+ CBCs were supplied by Bio-Resource Center .
- C34+ CBCs were cultured in hematopoietic culture medium [serum-free X-Vivo10 containing 50 ng/mL IL-6 , 50 ng/mL sIL-6 , 50 ng/mL SCF , ten ng/mL TPO , and 20 ng/mL Flt3/4 ligand ].
- Reprogrammed cells were cultured in feeder-less primate ES cell medium Repro FF (, .
- No. RCHEMD004), ReproFF2 (ReproCELL, cat No. RCHEMD006), mTeSR1 ( catalog number 05850) or E8 (16) supplemented with five ng/mL bFGF (total bFGF ten ng/mL) on Pronectin F-coated dishes.
- Passage of human iPSCs was previously described
Cell cultures
- Cord blood-derived mast cells were derived in supplemented medium'>StemPro-34 SFM medium including 100 ng/mL recombinant human SCF (hSCF , , ) and 10 ng/mL human IL-6 as previously described
Gene Delivery into CD34+ Cells by Retrovirus
- The CD34+ cells isolated from cord blood were cultured in X-VIVO15 , supplemented with 1% human serum albumin (HSA) and stimulated with a cytokine cocktail [100 ng/ml stem cell factor, 100 ng/ml Flt-3 ligand, 100 ng/ml thrombopoietin, and 100 ng/ml IL-6 ] in a 24-well plate (2×105 per well) for 48 h. The stimulated CD34+ cells were then harvested and placed into non-tissue culture-treated 6-well plates that had been coated with 20 µg/ml CH-296, a recombinant fibronectin fragment .
- (3×105 cells per well) in the presence of the respective virus supernatant.
- The virus supernatants were diluted 1∶2 with X-VIVO15 containing 1% HSA and the cytokine cocktail described above.
- Every 12 h, the medium was replaced with fresh virus supernatant.
- After 48 h of culture, the frequency of GFP- and/or Venus-expressing CD34+ cells was examined by FACS.
A human IgE preparation can induce IL-8 and IL-6 production and promote survival in human mast cells.
- Human CBMCs (1×105 cells) were incubated with 0 , 0.5, or 5 µg/mL of the indicated human IgEs in the absence of SCF and IL-6 for 72 hr.
Cell cultures
- Fresh UCB was obtained from , and the use of the UCB samples was reviewed and approved by the institutional review boards (IRB) of each cord blood collection hospital as well as those of the and National University of (NUS).
- The UCB-MNC were isolated using Ficoll Histopaque-1077 density-gradient centrifugation and counted before cyropreservation in 90% v/v donor autoplasma with 10% v/v dimethyl sulfoxide (DMSO ) for subsequent use.
- CD34+ selected cells were obtained using Magnetic Activated Cell Sorting (MACS) cell-separation columns (Milte-nyi Biotec GmbH, Bergisch Gladbach, Germany).
- The cryopreserved UCB-MNC were thawed using a thawing solution containing human albumin (HAS; 20% w/v , ) and Onkovertin 40 (10% w/v; B. Braun, ).
- The cells were centrifuged at 400
g for 15 min at 10 ° C. The cells were then washed with Dulbecco's phosphate-buffered saline , followed by centrifugation at 300g for another…